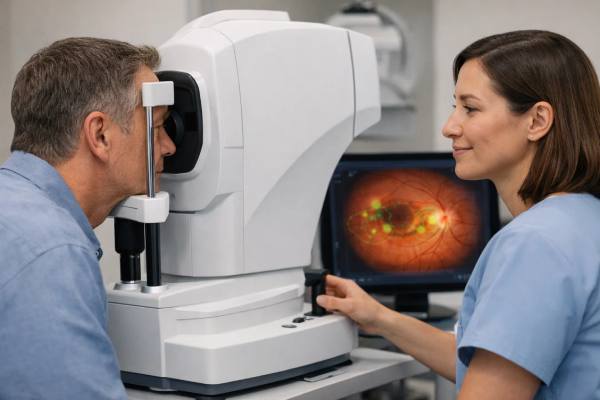
Retinal screening used to mean scheduling an eye specialist visit, dilating the pupils, and waiting for a human reader to interpret photos. New AI-enabled retinal scans compress that timeline. In many clinics, a trained staff member can capture high-quality images in minutes, and an algorithm can flag signs of disease immediately—often before symptoms appear. That speed matters because many vision-threatening conditions are silent until damage is advanced.
AI retinal screening is not a replacement for comprehensive eye care. It is best understood as an early-warning system: a way to find people who need timely follow-up, while helping others stay on track with routine checks. When used well, it can expand access, reduce missed disease, and standardize quality in busy settings. The most helpful approach is to know what the scan can detect, what it cannot, and how to act on results without overreacting.
Key Insights
- Earlier detection of diabetic retinopathy and other retinal findings can shorten the time to referral and treatment.
- Same-day results can improve screening adherence, especially when scans happen in primary care or community settings.
- False alarms and ungradable images are expected, so results should be paired with clear follow-up pathways.
- If you have diabetes, plan on screening at least yearly unless your eye clinician recommends a different interval.
Table of Contents
- What AI retinal screening actually is
- Which diseases are caught earlier
- How the scans and algorithms work
- Accuracy, false alarms, and blind spots
- What to expect at an AI scan visit
- Who benefits most and when to repeat
- Privacy, fairness, and oversight in clinics
What AI retinal screening actually is
AI retinal screening combines two things: a fast way to capture images of the back of the eye, and a software system trained to detect patterns linked to disease. The images may be standard color photographs of the retina, or scans such as optical coherence tomography, which maps retinal layers in cross-section. In many programs, the image capture is done without dilation using a non-mydriatic fundus camera, which makes the process simpler and more comfortable.
It helps to separate three related terms that are often blended together:
- Retinal imaging: the camera or scanner takes images. Imaging by itself does not diagnose anything.
- AI decision support: an algorithm offers a probability score or highlights suspicious areas, but a clinician still makes the official interpretation.
- Autonomous AI screening: the system produces a clinical output without a clinician reading the image at the point of care, usually as “refer” or “no refer,” with rules about when an image is too poor to grade.
In real-world workflows, AI screening is primarily a triage tool. The main aim is not to describe every tiny finding. It is to identify people who may have meaningful disease and need a timely, higher-resolution evaluation by an eye professional.
A practical way to understand the value is to look at the current bottleneck. Many conditions require regular screening, but attendance is inconsistent, and eye specialist capacity is limited. AI programs are designed to shift basic screening closer to where patients already are, such as primary care clinics, diabetes clinics, pharmacies, or mobile units. The most successful models treat the scan as part of routine care rather than a separate, hard-to-schedule appointment.
When patients worry that “AI is diagnosing me,” the most accurate framing is this: the AI is classifying the image against a well-defined target, usually a referral threshold. If the system flags you, it means you likely need a full eye evaluation soon. If it does not flag you, it does not guarantee you are disease-free. It means the algorithm did not detect the specific patterns it was trained to find at that threshold, on the images it received.
Which diseases are caught earlier
The retina is one of the few places in the body where clinicians can directly view small blood vessels and living nerve tissue without surgery. That is why retinal screening has always been valuable. AI strengthens that value by improving consistency and making screening easier to deliver at scale.
Diabetic retinopathy and diabetic macular edema
The strongest evidence and the most widespread deployment are in diabetic retinopathy screening. Early diabetic retinopathy can be symptom-free, and delayed detection can mean avoidable vision loss. AI systems are commonly trained to detect “referable” disease, which roughly means changes severe enough to warrant evaluation by an eye clinician, and in some systems, signs of diabetic macular edema risk.
Earlier detection here is not only about finding disease sooner in an individual. It is also about catching disease in more people overall. When screening is available where people already receive diabetes care, fewer patients fall through the cracks.
Age-related macular degeneration and macular warning signs
Some AI tools analyze retinal photos for drusen patterns and pigment changes that suggest early age-related macular degeneration. Others focus on optical coherence tomography findings. Earlier detection may help patients start monitoring strategies sooner, address modifiable risks, and ensure timely evaluation if symptoms develop. It can also reduce the delay between “something feels off” and “a scan shows fluid,” which is critical for treatable wet forms of macular disease.
Glaucoma risk signals
Glaucoma is primarily diagnosed using optic nerve assessment, eye pressure, and functional testing like visual fields. Retinal photos can show optic nerve head changes and nerve fiber layer clues, and OCT can quantify nerve fiber thickness. AI can flag suspicious optic nerve features that deserve comprehensive glaucoma evaluation. It does not replace a full glaucoma workup, but it can identify people who might otherwise go untested until later stages.
Hypertensive retinopathy and vascular clues
Retinal vessel changes can reflect chronic high blood pressure and other vascular risks. While not all programs report these findings clinically, the concept matters: retinal images can reveal systemic health signals. In settings where eye and primary care are integrated, this can prompt earlier discussion about blood pressure control and cardiovascular risk assessment.
Incidental findings that still matter
Even when a scan is designed for a specific disease, the images may reveal other concerns: retinal vein occlusions, suspicious pigmentation, or signs of past inflammation. Some workflows include a safety net where abnormal findings trigger referral even if they are outside the algorithm’s core target. The key is transparency about what the program promises to detect and what it does not.
How the scans and algorithms work
AI retinal screening is built on two foundations: image quality and model training. If either is weak, results become less reliable.
The imaging step: what the camera is really capturing
Most screening programs use a non-mydriatic fundus camera that captures one or more fields of the retina through an undilated pupil. This is fast, but it depends on cooperation, tear film clarity, and adequate pupil size. Cataracts, dry eye, small pupils, and poor fixation can all reduce image quality. Some systems provide real-time feedback to the operator, prompting them to retake images or adjust alignment.
Optical coherence tomography is different. It uses reflected light to produce cross-sectional “slices” of the retina, making swelling, fluid, and layer changes easier to see. OCT can be powerful for macular disease and glaucoma-related structural assessment, but it is typically more expensive and may be less common in mass screening settings.
How algorithms learn patterns
Most modern retinal screening models are based on deep learning. They are trained on large datasets of labeled images, where the labels come from expert readers, reading centers, or reference standards that may include additional imaging. During training, the model learns statistical relationships between pixel patterns and the label categories.
Two details are crucial for understanding performance:
- The reference standard defines the task. If the label is “referable diabetic retinopathy,” the model learns that threshold. It is not learning “all possible retinal disease.”
- The training population shapes generalization. If images mostly come from certain cameras, lighting conditions, and patient demographics, performance can drift in other settings.
Outputs are often simpler than people expect
Many clinically deployed systems output a limited set of results, such as:
- “More than mild diabetic retinopathy detected” versus “not detected”
- “Vision-threatening diabetic retinopathy risk” versus “not detected”
- “Ungradable” or “insufficient image quality”
That simplicity is intentional. Screening works best when it produces an actionable next step rather than a confusing set of probabilities. When probability scores are provided, they should be paired with clear thresholds and an explanation of uncertainty.
Why “immediate” results can still be safe
Same-day output does not mean uncontrolled automation. Well-designed systems use locked models, quality checks, and conservative referral thresholds. They also build in “fail safely” rules, such as sending ungradable images for in-person evaluation rather than issuing a false reassurance.
A helpful mental model is airport security: the scanner does not prove you are safe, but it is designed to detect patterns that warrant further inspection, quickly and consistently.
Accuracy, false alarms, and blind spots
People often ask, “How accurate is AI retinal screening?” The most honest answer is, “Accurate within a defined job, with predictable trade-offs.” Screening systems are built to minimize missed disease while keeping false alarms at a manageable level.
Sensitivity and specificity in plain language
- Sensitivity is how well the system catches people who truly have the target disease at the referral threshold. Higher sensitivity means fewer missed cases.
- Specificity is how well it avoids flagging people who do not have that disease. Higher specificity means fewer unnecessary referrals.
In screening, sensitivity is often prioritized because missing treatable disease is costly. That means some false positives are expected, especially when disease prevalence is low in the screened population.
Why false positives are not always “errors”
A flagged result can happen for several reasons:
- The algorithm detected subtle patterns that genuinely deserve confirmation.
- The image quality was borderline, and the system leaned toward safety.
- The model picked up look-alike changes such as old laser scars, pigment changes, or artifacts.
In practice, the “cost” of a false positive is a follow-up exam that ultimately reassures you. That cost is real, but it is usually acceptable when balanced against the benefit of catching true disease earlier.
The ungradable problem
A common output in real-world screening is “ungradable” or “insufficient image quality.” This is not a failure; it is the system admitting uncertainty. Ungradable rates vary by setting, camera, operator experience, and patient factors like cataract and small pupils. Programs should treat ungradable images as clinically meaningful because poor imageability can correlate with media opacity and older age, both of which can increase eye disease risk.
If you receive an ungradable result, the next step is usually a dilated eye exam or repeat imaging with a different approach.
Blind spots to respect
Even excellent AI screening systems have limitations:
- They may not detect diseases outside their training targets, such as retinal tears, uveitis, or subtle tumors.
- They may perform less reliably in unusual anatomy, extreme myopia, or after certain surgeries.
- Performance can vary across devices and populations if the model was not validated broadly.
- They do not replace symptom-based care. New floaters, flashes, distortion, or sudden vision changes still require prompt evaluation even if a screening result was normal.
The safest expectation is this: AI screening can meaningfully reduce missed disease at a population level, but it does not eliminate the need for comprehensive exams and symptom-driven eye care.
What to expect at an AI scan visit
For many people, the first AI retinal scan happens in a setting that does not look like an eye clinic. Knowing the steps can reduce anxiety and improve image quality.
Before the scan
You will usually be asked about eye history and diabetes status, and sometimes about prior retinopathy. If you have dry eye, using lubricating drops 10–15 minutes beforehand can improve image clarity. Avoid rushing in from bright sunlight, since pupil size affects image capture; a few minutes indoors can help.
If you wear contact lenses, most fundus photography can be done with lenses in place, but if your eyes feel dry or your vision is hazy, removing them may improve image quality.
During image capture
A staff member will position you at the camera, and you will look at a fixation target. The camera may flash. The whole process often takes less than five minutes once aligned, though retakes can add time.
Some programs use single-field imaging; others capture multiple fields per eye. More fields can improve detection of peripheral changes, but it may take longer and require more cooperation.
Results and what they mean
Outputs are typically structured so the next step is obvious. Common categories include:
- No referable disease detected: generally means routine follow-up at the recommended interval, unless you have symptoms or known disease that requires closer monitoring.
- Refer or positive screen: means schedule a comprehensive eye exam within a specified timeframe, often weeks rather than months, depending on severity category.
- Ungradable: means the images were not sufficient to make a safe call, and you should arrange an eye exam or repeat imaging.
A high-quality program will give you clear written instructions that match the result category. If you leave with a vague message like “AI found something,” ask for the exact output category and recommended timeframe.
How clinicians act on results
Strong workflows do not stop at generating a result. They include:
- A referral pathway to eye care providers who can see patients promptly
- A system for tracking whether follow-up occurred
- A plan for repeat attempts if images are poor
- A strategy for high-risk individuals, such as those with long-standing diabetes
If your clinic offers AI screening, it is reasonable to ask how follow-up is coordinated. The scan is only as useful as the next step.
Who benefits most and when to repeat
AI retinal screening is most valuable when it reaches people who are least likely to get traditional eye exams on schedule. It can also help standardize care for large health systems where screening volume is high.
High-benefit groups
AI screening tends to be especially helpful for:
- People with diabetes who miss yearly eye exams: same-day screening in primary care can remove scheduling friction.
- Communities with limited eye specialist access: mobile or community-based programs can find disease earlier and reduce travel barriers.
- Patients who need frequent monitoring but have competing priorities: screening integrated into routine care can keep follow-up from slipping.
- Older adults with multiple health appointments: combining screening with existing visits reduces the burden of additional appointments.
It can also be useful for patients with transportation barriers, caregiving responsibilities, or work schedules that make specialty visits difficult.
Suggested repeat intervals
Screening intervals should be individualized, but there are practical starting points:
- Diabetes: many adults are advised to have retinal screening at least annually unless an eye clinician recommends a different schedule based on prior findings, pregnancy, glycemic control, and other risk factors.
- Known retinopathy: once disease is present, screening often shifts from “screening” to “monitoring,” and intervals may tighten. AI screening may still be used in some settings, but specialist oversight becomes more important.
- Glaucoma suspicion or high risk: screening flags should lead to a comprehensive glaucoma evaluation. Ongoing follow-up is typically guided by optic nerve status, pressure, and visual field testing.
A key point is that a “normal” AI screen does not reset risk to zero. It simply means no target-level findings were detected at that time, on those images. Risk continues to accumulate with age, duration of diabetes, blood pressure, and other factors.
How to use results wisely
If your result is normal, the best next step is to treat it as confirmation that you are on track, not as a reason to disengage. Keep the interval consistent. If your result is positive, focus on timing and follow-through rather than panic. The value of screening is not the label; it is the path to confirmation and, when needed, treatment.
If you want a simple rule: use AI screening to reduce missed disease, not to replace your relationship with an eye clinician when risk is high or symptoms are present.
Privacy, fairness, and oversight in clinics
Retinal images are medical data. They can also be uniquely identifying because blood vessel patterns are distinctive. That makes privacy, governance, and fairness central to responsible AI screening.
Data handling and consent
A well-run program should be clear about:
- Where images are stored: on-site, in a secure cloud, or within an electronic health record system
- Who can access them: clinical staff, eye specialists, and authorized program administrators
- How long they are retained: retention often follows medical record policies
- Whether images are used to improve models: some programs use de-identified images for quality improvement or future training, which should be disclosed
If you are uncomfortable, ask whether you can opt out of secondary use while still receiving clinical screening.
Bias and generalizability
AI systems can underperform in populations underrepresented in training data. In retinal imaging, this can show up as differences in imageability, camera performance, and disease presentation across groups. Responsible programs address this by:
- Validating performance across diverse patient populations
- Monitoring outcomes over time, not just at launch
- Tracking ungradable rates by site and demographic factors
- Updating workflows when disparities appear, such as offering dilation or alternative imaging when needed
Fairness is not only about the model. It is also about access to follow-up. A positive screen that does not lead to timely specialist care can widen disparities rather than reduce them.
Clinical oversight and accountability
AI screening should have clear lines of responsibility. Patients should know:
- Who to contact with questions about the result
- How quickly follow-up is expected
- What happens if images are ungradable
- Whether a clinician reviews outputs in certain scenarios
Clinics should also run quality audits. Even with a locked model, real-world performance can drift due to camera replacement, staffing changes, or shifts in patient mix. The most mature programs treat AI as a clinical tool that needs ongoing measurement, not as a one-time installation.
The healthiest mindset is cautious optimism: AI retinal screening can be genuinely transformative when paired with strong governance, clear communication, and reliable follow-up care.
References
- The efficacy of artificial intelligence in diabetic retinopathy screening: a systematic review and meta-analysis 2025 (Systematic Review and Meta-Analysis)
- Diabetic Retinopathy Preferred Practice Pattern® 2025 (Guideline)
- Pivotal Evaluation of an Artificial Intelligence System for Autonomous Detection of Referrable and Vision-Threatening Diabetic Retinopathy 2021 (Prospective Diagnostic Study)
- Deep Learning in Glaucoma Detection and Progression Prediction: A Systematic Review and Meta-Analysis 2025 (Systematic Review and Meta-Analysis)
- A SYSTEMATIC REVIEW OF DEEP LEARNING APPLICATIONS FOR OPTICAL COHERENCE TOMOGRAPHY IN AGE-RELATED MACULAR DEGENERATION 2022 (Systematic Review)
Disclaimer
This article is for educational purposes and does not provide medical advice. AI retinal screening can help identify people who need timely eye evaluation, but it does not replace a comprehensive eye exam or urgent assessment for new symptoms. Seek prompt care for sudden vision loss, new flashes or floaters, eye pain, significant light sensitivity, or distortion of straight lines. If you receive a positive or ungradable screening result, follow the recommended timeframe for a full eye evaluation, since only a clinician can confirm a diagnosis and determine the right treatment plan.
If this article was useful, please consider sharing it on Facebook, X (formerly Twitter), or any platform you prefer.