Bacterial endocarditis (often called infective endocarditis) is a serious infection of the heart’s inner lining, usually involving one or more heart valves. It starts when bacteria enter the bloodstream—sometimes from the mouth, skin, gut, urinary tract, or an IV line—and then latch onto a rough spot inside the heart. There, they can build “vegetations,” clumps of bacteria, platelets, and inflammatory debris that damage valves and can break off like tiny splinters, traveling to the brain, lungs, kidneys, or spleen. The condition can develop quickly over days or smolder for weeks, which is why it is sometimes missed early. With timely blood cultures, imaging, and the right antibiotics (and surgery when needed), many people recover—but delays raise the risk of stroke, heart failure, and long-term valve problems.
Table of Contents
- What it is and why it is dangerous
- What causes it and who is at risk
- Early symptoms and common complications
- How doctors diagnose bacterial endocarditis
- Treatments that work and what to expect
- Management, prevention and when to seek urgent care
What it is and why it is dangerous
Bacterial endocarditis is an infection on the inside surface of the heart—most often on the heart valves, but sometimes on implanted material (a prosthetic valve, repair ring, pacemaker lead) or on damaged endocardium. The hallmark is a vegetation: bacteria embedded in a sticky scaffold of platelets and fibrin. This matters for two reasons. First, vegetations are hard for the immune system to clear, so the infection tends to persist unless treated with high-dose antibiotics for weeks. Second, vegetations can erode valve tissue or break apart and shower infected fragments into the bloodstream.
The way the illness behaves often reflects the organism and the patient’s heart. Some cases are acute, with high fever and rapid decline—classically seen with aggressive bacteria such as Staphylococcus aureus. Others are subacute, with milder fever and weeks of fatigue, weight loss, or night sweats—often linked to oral streptococci after dental disease or gum bleeding. The “acute vs subacute” idea is helpful, but real life can be mixed: older adults may not spike high fevers, and people taking anti-inflammatory medicines may mask symptoms.
Damage can be local (valve leakage, abscesses around the valve ring, heart rhythm problems) or systemic (stroke, kidney inflammation, lung infections in right-sided disease). Even with modern care, infective endocarditis has meaningful mortality; published estimates often place one-year mortality around 30% to 40% in many populations, largely due to complications and underlying illness.
A useful mental model is this: endocarditis is not “just” an infection—it is an infection plus a mechanical heart problem. Successful treatment often requires both microbiology precision and cardiology-level planning.
What causes it and who is at risk
Endocarditis usually requires two things: bacteria in the bloodstream (bacteremia) and a surface inside the heart that allows bacteria to stick. Bacteremia can happen from everyday activities—brushing teeth with inflamed gums can cause brief bacteremia—yet most people never develop endocarditis because their valves and endocardium are smooth and resistant.
Common bacterial culprits include:
- Staphylococcus aureus (including MRSA): often linked to skin sources, IV drug use, and IV lines; tends to be aggressive.
- Viridans group streptococci: commonly associated with the mouth, dental disease, and some dental procedures.
- Enterococcus faecalis and related enterococci: can originate from the gastrointestinal or urinary tract, especially in older adults.
- Coagulase-negative staphylococci: more common with prosthetic valves or implanted cardiac devices.
- HACEK organisms: a smaller group from the mouth and upper airway; less common but well-known in classic teaching.
Risk rises when blood flow is turbulent or foreign material is present. Higher-risk situations include:
- Prosthetic heart valves (surgical or transcatheter) or prior valve repair with prosthetic material
- Previous infective endocarditis
- Certain congenital heart diseases (especially cyanotic lesions or repairs with residual defects)
- Cardiac implantable electronic devices (pacemakers, ICDs) or vascular grafts
- Hemodialysis and frequent vascular access
- Injection drug use (particularly right-sided, involving the tricuspid valve)
- Long-term IV catheters, including PICC lines or ports
- Poor oral health (periodontal disease, untreated dental infections)
- Immunosuppression (for example, chemotherapy or high-dose steroids)
Triggers are often not dramatic. A dental abscess, a skin boil, a urinary infection, or a contaminated IV line can be enough. Sometimes no clear entry point is found, which is why clinicians focus on identifying the organism and evaluating the heart rather than relying on a single presumed source.
Early symptoms and common complications
Symptoms can be deceptively nonspecific early on. Many people describe “a flu that will not go away,” especially in subacute cases. The most common early symptoms include:
- Fever or chills (but some older adults have normal temperature)
- Fatigue, weakness, reduced exercise tolerance
- Night sweats
- Loss of appetite and weight loss
- Shortness of breath or swelling in the legs if valve leakage develops
- New or changing heart murmur (not always present or noticed)
Clues that should raise suspicion—especially in someone with risk factors—include persistent fever for more than a few days, fevers plus a new heart symptom, or fevers plus signs of emboli (below).
Classic physical findings exist, but they are less common than medical textbooks suggest:
- Petechiae (tiny red or purple spots) on skin or inside the mouth
- Splinter hemorrhages under nails
- Painful finger or toe nodules (Osler nodes) and painless palm or sole lesions (Janeway lesions)
- Eye findings such as retinal hemorrhages
Complications are the reason endocarditis is treated so urgently:
- Heart failure from severe valve regurgitation (leakage) or valve obstruction.
- Stroke and brain complications from emboli; symptoms can be sudden weakness, speech trouble, facial droop, severe headache, or confusion.
- Abscess formation around the valve ring (perivalvular abscess), which can cause persistent fever despite antibiotics and may disrupt electrical conduction, leading to new heart block.
- Septic emboli to other organs:
- Kidneys: blood in urine, rising creatinine, inflammatory kidney injury
- Spleen: left upper abdominal pain or tenderness
- Lungs (more typical in right-sided disease): pleuritic chest pain, cough, shortness of breath, cavitating lesions
- Persistent bloodstream infection and sepsis, especially with S. aureus.
If you take away one practical point: endocarditis symptoms can be quiet while complications are loud. New neurologic symptoms, chest pain, fainting, or rapidly worsening breathlessness should be treated as emergencies.
How doctors diagnose bacterial endocarditis
Diagnosis relies on combining microbiology, imaging, and clinical clues. Because antibiotics can sterilize the bloodstream quickly, the most important early step—when the patient is stable—is usually blood cultures before antibiotics.
Typical diagnostic steps include:
- Blood cultures
- Many clinicians obtain at least three sets from separate draws, ideally before the first antibiotic dose.
- Persistent positive cultures (especially with organisms known to cause endocarditis) strongly support the diagnosis.
- If cultures stay negative but suspicion remains high, teams consider prior antibiotic exposure and order specialized tests (for example, serology or molecular tests for harder-to-grow pathogens).
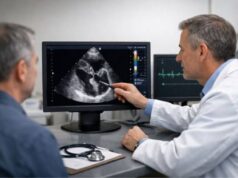
- Echocardiography (heart ultrasound)
- Transthoracic echo (TTE) is noninvasive and often the first test.
- Transesophageal echo (TEE) places the probe in the esophagus and provides sharper images; it is especially valuable for prosthetic valves, device leads, abscesses, or when TTE is nondiagnostic.
- Echo can show vegetations, valve perforation, severe leakage, abscess cavities, and prosthetic valve problems.
- Diagnostic criteria
- Clinicians commonly use structured criteria (often based on Duke-style frameworks) that weigh:
- Major evidence: typical organisms in blood cultures, clear imaging findings
- Minor evidence: fever, predisposing heart conditions, vascular or immune phenomena
- Advanced imaging and complication checks
- Depending on the case, the team may look for “silent” emboli or abscesses with brain imaging, CT of chest and abdomen, or other targeted studies.
- In prosthetic valve or device-related cases, additional imaging modalities can help clarify whether infection is present when echo images are limited.
- Baseline labs and monitoring
- CBC, kidney and liver tests, inflammatory markers, urinalysis, and ECG are common.
- These help track treatment response and detect complications such as kidney injury, anemia, or conduction abnormalities.
A practical reality: diagnosis is often a race between “prove it” and “treat it.” If a patient is unstable—septic shock, severe respiratory distress, or neurologic catastrophe—clinicians may start antibiotics immediately while still drawing cultures as quickly as possible.
Treatments that work and what to expect
Treatment has two main goals: eradicate the bacteria and prevent or repair structural heart damage. Most patients need hospitalization at least initially.
1) Antibiotics (the core therapy)
Endocarditis bacteria are protected inside vegetations, so therapy is typically:
- High-dose, targeted IV antibiotics
- For 4 to 6 weeks in many cases (sometimes longer for prosthetic valves or complicated infections)
- Adjusted to the organism and its antibiotic susceptibility profile
Clinicians often start empiric therapy (broad coverage) after blood cultures are drawn, then narrow to the most effective regimen once results return. Because dosing and combinations vary widely, plans are individualized—especially for MRSA, enterococci, kidney disease, or drug allergies.
In selected stable patients who respond well, some programs use outpatient parenteral antibiotic therapy (OPAT) with close monitoring. In specific situations and under specialist guidance, a carefully chosen oral step-down approach may be considered after an initial IV phase, but it is not appropriate for everyone and depends on organism, complications, adherence, gut absorption, and follow-up reliability.
2) Surgery (when antibiotics are not enough)
Surgery is not rare in endocarditis because antibiotics cannot fix a valve that is torn, leaking severely, or surrounded by an abscess. Common reasons include:
- Heart failure from acute severe valve regurgitation or obstruction
- Uncontrolled infection, such as persistent bacteremia despite appropriate antibiotics
- Abscesses or destructive valve complications
- Recurrent emboli or very large vegetations with high embolic risk
- Prosthetic valve dysfunction or dehiscence
- Infected device leads (often requiring complete system removal)
Timing is case-specific. Sometimes surgery is emergent (for cardiogenic shock). Other times, the team stabilizes infection first. Neurologic events complicate timing decisions and require coordination among cardiology, infectious diseases, neurology, and cardiac surgery.
3) Supportive care and monitoring
During therapy, clinicians monitor:
- Fever curve and repeat cultures (to confirm bloodstream clearance)
- Kidney function and antibiotic levels when needed
- ECG changes (new conduction disease can hint at abscess)
- Signs of emboli or worsening valve function
What patients often experience: a slow return of energy, frequent blood tests, possible line placement (PICC), and a structured follow-up plan after discharge.
Management, prevention and when to seek urgent care
Recovery does not end when antibiotics stop. The weeks after treatment are when people rebuild stamina, manage medication effects, and watch for relapse or valve problems.
Day-to-day management during and after treatment
- Take antibiotics exactly as prescribed. Missed doses increase relapse risk.
- Protect IV access. Keep dressings clean and dry, wash hands before handling tubing, and report redness, drainage, fever, or line pain promptly.
- Track symptoms. A simple daily log (temperature, weight, swelling, shortness of breath, new pain) can catch trouble early.
- Follow monitoring plans. Lab checks are not “busywork”—they help prevent kidney injury and catch side effects.
- Rebuild activity gradually. Many people feel deconditioned. Short walks, gentle pacing, and stepwise goals often work better than sudden exertion.
- Plan dental care thoughtfully. Oral health is a long-term protective factor: regular cleanings, gum care, and treatment of dental infections reduce everyday bacteremia.
Prevention: what actually helps
- Excellent oral hygiene (daily brushing, flossing if tolerated, management of gum disease)
- Skin and wound care (prompt treatment of abscesses, avoid picking at skin lesions)
- Safe injection practices and support for substance use disorder, if relevant (this can be the most impactful prevention step for right-sided disease)
- Careful handling of catheters and dialysis access
Antibiotic prophylaxis before dental procedures
Some people at highest risk of severe outcomes may be advised to take a single antibiotic dose before certain dental procedures that manipulate gums or the tooth root area. High-risk groups often include those with prosthetic valves, prior endocarditis, and select congenital heart diseases. The exact recommendation, antibiotic choice, and dose depend on local guidance and allergy history, and should be confirmed with the treating clinician—especially because some older regimens are no longer favored due to side effects.
When to seek urgent care (do not wait)
Go to emergency services or contact an urgent clinician if any of the following occur during treatment or in the months afterward:
- Sudden weakness, numbness, facial droop, speech difficulty, severe headache, confusion
- New chest pain, fainting, or severe shortness of breath
- Persistent fever (for example, ≥38°C / 100.4°F) after initial improvement
- New or worsening leg swelling, rapid weight gain over 1 to 2 days
- Signs of sepsis: shaking chills, very low blood pressure symptoms, extreme drowsiness
- IV line redness, drainage, severe pain, or swelling
Prevention and early response are not about fear—they are about respecting a condition that can change quickly and treating small warning signs as valuable data.
References
- 2023 ESC Guidelines for the management of endocarditis 2023 (Guideline)
- The 2023 Duke-International Society for Cardiovascular Infectious Diseases Criteria for Infective Endocarditis: Updating the Modified Duke Criteria 2023 (Diagnostic Criteria)
- Guidelines for Diagnosis and Management of Infective Endocarditis in Adults: A WikiGuidelines Group Consensus Statement 2023 (Guideline and Systematic Review)
- Antibiotic Prophylaxis and Infective Endocarditis Incidence Following Invasive Dental Procedures: A Systematic Review and Meta-Analysis 2024 (Systematic Review)
Disclaimer
This article is for general educational purposes and is not a substitute for personal medical advice, diagnosis, or treatment. Bacterial endocarditis can be life-threatening and requires prompt, individualized care from qualified clinicians. If you think you may have endocarditis—especially if you have a prosthetic valve, a cardiac device, or persistent fever—seek urgent medical evaluation. Never start, stop, or change antibiotics or blood-thinning medicines based on an article, and always follow the plan set by your cardiology and infectious diseases team.
If you found this helpful, please consider sharing it on Facebook, X (formerly Twitter), or any platform you prefer, and follow us on social media. Your support through sharing helps our team keep producing reliable, high-quality health content.