Cardiac allograft vasculopathy (CAV) is a form of coronary artery disease that develops only in transplanted hearts. Instead of creating a few isolated blockages, it often causes a diffuse, concentric narrowing along long segments of the coronary arteries and can also involve the small vessels that do not show up well on standard angiograms. That pattern matters: it can reduce blood flow quietly for years, then present as heart failure, arrhythmia, or sudden decline.
CAV is best understood as chronic injury to the graft’s blood vessel lining—part immune, part metabolic, part infectious, and part mechanical. Because the transplanted heart is often denervated, classic angina may be absent, so structured surveillance is central to staying ahead of the disease. This article explains what CAV is, why it happens, how risk is shaped, what symptoms to watch for, how clinicians diagnose and stage it, and how treatment and daily management decisions are made after heart transplantation.
Table of Contents
- What CAV is and why it looks different
- What causes CAV after heart transplant
- Risk factors that raise progression risk
- Symptoms and complications to watch for
- How CAV is diagnosed and monitored
- Treatment and long-term management
What CAV is and why it looks different
Cardiac allograft vasculopathy is a progressive disease of the coronary circulation in a transplanted heart. Its hallmark is uniform thickening of the inner vessel layer (intimal hyperplasia) that tends to involve long segments of the artery rather than forming a few discrete plaques. Clinically, this matters because “spot fixes” (like opening one short blockage) may not restore normal perfusion if the entire vessel tree is gradually narrowing.
Two features make CAV different from typical coronary artery disease:
- Diffuse involvement: Narrowing may extend from the proximal to distal branches, reducing the heart’s ability to increase blood flow during exertion. Even “mild” narrowing can be significant if it is widespread.
- Microvascular disease: Small-vessel dysfunction can coexist with epicardial disease. A patient can have a “normal-looking” angiogram and still have impaired perfusion reserve, especially early in the disease.
CAV can also be clinically stealthy. Many transplanted hearts are denervated, which reduces the likelihood of classic chest pain. Instead, the heart may signal trouble through:
- reduced exercise capacity
- shortness of breath
- unexplained fatigue
- fluid retention
- arrhythmias or conduction abnormalities
Another important difference is time course. CAV can begin early (sometimes within the first year) as endothelial injury accumulates, then accelerate in the setting of rejection episodes, donor-specific antibodies, or uncontrolled metabolic risk. It is often described as a form of chronic rejection, but that label is incomplete. In reality, CAV sits at the intersection of immune injury, infection-related inflammation, and traditional cardiovascular risk factors—plus donor and procedural contributors such as ischemia-reperfusion injury.
Clinicians typically think of CAV as a staged problem: early endothelial dysfunction and intimal thickening, then progressively reduced flow reserve, then clinically apparent ischemia, graft dysfunction, and complications. This staging approach shapes surveillance plans and treatment choices. The goal is not simply to “find stenosis,” but to identify when CAV is beginning to affect perfusion or ventricular function so therapy can be intensified before irreversible injury occurs.
What causes CAV after heart transplant
CAV develops from repeated injury to the coronary endothelium (the inner lining of blood vessels) followed by maladaptive repair. That repair process becomes self-reinforcing: inflammation promotes smooth-muscle migration and collagen deposition, which thickens the vessel wall, narrows the lumen, and further stresses the endothelium.
It helps to group causes into immune and non-immune drivers, while remembering that most patients have both.
Immune-driven mechanisms
Immune injury can occur even when routine symptoms are minimal. Important immune contributors include:
- Cellular rejection episodes: Each episode can inflame the graft vasculature and accelerate remodeling, particularly when rejection is recurrent or severe.
- Antibody-mediated rejection and donor-specific antibodies: Antibodies can bind vascular targets and activate complement and inflammation, creating persistent endothelial stress.
- Chronic low-grade immune activation: Even without overt rejection, subclinical immune activity may continue to irritate the vasculature over time.
Non-immune mechanisms
Non-immune contributors often look like the same risks that drive native coronary disease—but they interact with transplant-specific biology:
- Ischemia-reperfusion injury: The transplant process itself can injure the endothelium, making vessels more vulnerable to later immune and metabolic hits.
- Cytomegalovirus and other infections: Certain infections can amplify inflammation and endothelial activation, and infection control is often treated as part of CAV prevention.
- Metabolic effects of immunosuppression: Some regimens increase blood pressure, lipids, blood glucose, and weight, each of which can accelerate vascular injury.
- Traditional risk factors: Hypertension, dyslipidemia, diabetes, smoking exposure, obesity, and sedentary behavior all contribute to endothelial dysfunction and plaque biology.
- Donor and graft characteristics: Donor age and pre-existing donor coronary disease can increase baseline risk, and later diffuse changes may compound an already vulnerable system.
A useful mental model is “injury plus response.” The injury can be immune (rejection, antibodies), infectious (viral activation), or metabolic (lipids, glucose, blood pressure). The response is a vessel-wall remodeling program that is unusually diffuse and proliferative in transplanted hearts.
One practical insight: many patients feel well while CAV is developing, so clinicians rely on structured surveillance to detect early disease. That surveillance is not just precaution—it reflects the biology of CAV, which can advance silently until it reaches a threshold where graft function drops quickly.
Risk factors that raise progression risk
Not every heart transplant recipient develops clinically important CAV, and progression rates vary. Risk is shaped by the interaction of donor factors, recipient biology, immune history, infection burden, and long-term control of vascular risk factors. Thinking in categories can help patients and clinicians target what is modifiable.
Donor and transplant-related risk factors
These influence the graft’s baseline vulnerability:
- Older donor age
- Donor coronary artery disease (recognized or unrecognized)
- Longer ischemic time and ischemia-reperfusion injury
- Early graft dysfunction, which can reflect global endothelial stress
Immune and rejection-related risk factors
These are among the strongest predictors of progression:
- Recurrent acute cellular rejection
- Antibody-mediated rejection
- Persistent or rising donor-specific antibodies
- Graft inflammation that requires intensification of immunosuppression
Infection and inflammation-related risk factors
Certain infections appear to increase endothelial activation and vascular remodeling:
- Cytomegalovirus infection or reactivation
- Recurrent systemic infections, especially early after transplant when immune balance is most delicate
- Chronic inflammatory states that keep cytokines elevated
Traditional cardiovascular risk factors in transplant recipients
These are often treatable and are central to long-term care:
- Elevated LDL cholesterol and triglycerides
- Hypertension
- Diabetes or impaired glucose tolerance
- Chronic kidney disease (which amplifies vascular calcification and endothelial dysfunction)
- Smoking exposure
- Obesity and low cardiorespiratory fitness
A key nuance is that “traditional” risks can be amplified by immunosuppression. For example, calcineurin inhibitors can raise blood pressure and worsen kidney function; corticosteroids can worsen glucose control and weight. This is why transplant programs often start vascular prevention early and monitor metabolic markers aggressively rather than waiting for problems to declare themselves.
Who tends to progress faster in real practice
While individual prediction is imperfect, faster progression is often seen in:
- Patients with repeated rejection or antibody activity despite optimized therapy
- Patients with uncontrolled lipids, blood pressure, or diabetes over several years
- Patients with significant kidney disease and high medication complexity
- Patients with early signs of microvascular dysfunction or reduced flow reserve even when angiography appears mild
The most actionable takeaway is that CAV prevention is rarely one single intervention. It is a long-term “stack” of protective steps: consistent immunosuppression adherence, early rejection detection, infection prevention, and tight control of lipids, blood pressure, and glucose. Small lapses across multiple areas can add up to meaningful vascular injury over time.
Symptoms and complications to watch for
CAV is often asymptomatic early because the transplanted heart frequently lacks normal pain signaling. As a result, symptoms tend to be indirect signs of impaired perfusion, rising filling pressures, or rhythm instability. Many patients also have overlapping problems—kidney disease, anemia, deconditioning, medication side effects—so symptom interpretation is most reliable when paired with objective testing.
Common symptoms when CAV becomes clinically relevant
- Reduced exercise tolerance: Needing more breaks, slower pace, fewer stairs, longer recovery time
- Shortness of breath with exertion: Often gradual, sometimes sudden if disease accelerates
- Unexplained fatigue: Especially when it is new or progressively worsening
- Palpitations or irregular heartbeat: Can signal atrial arrhythmias or ventricular irritability
- Fluid retention: Swelling in legs or abdomen, rapid weight gain, or needing higher diuretic doses
- Atypical chest discomfort: Some recipients do feel chest pressure, but the absence of pain does not exclude significant ischemia
Why angina may be absent
After transplant, cardiac nerves may be partially or fully disrupted. Even when some reinnervation occurs years later, the pattern is variable. This means ischemia can show up as:
- shortness of breath rather than pain
- nausea, sweating, or sudden fatigue
- syncope or near-syncope in advanced disease
- “silent” changes detected only on surveillance testing
Major complications
CAV can lead to several serious outcomes:
- Graft dysfunction: Both systolic and diastolic function can decline as chronic ischemia affects the myocardium.
- Heart failure episodes: Fluid overload and hospitalizations may become more frequent.
- Arrhythmias and conduction disease: Ischemic scarring and microvascular injury can trigger atrial fibrillation, ventricular arrhythmias, or heart block.
- Myocardial infarction: Less common as a classic single-lesion event, but possible, particularly with focal disease superimposed on diffuse narrowing.
- Sudden cardiac death: A feared late complication, often related to malignant arrhythmias or severe ischemia.
Symptoms that should prompt urgent evaluation
Seek urgent medical care if any of the following occur:
- fainting, near-fainting, or new severe lightheadedness
- sudden or rapidly worsening shortness of breath at rest
- new chest pressure at rest or with minimal activity
- rapid weight gain with swelling and reduced urine output
- palpitations plus dizziness, chest discomfort, or breathlessness
A practical tracking approach
Because gradual change is easy to miss, many transplant teams encourage simple tracking:
- a consistent walking route and time to complete it
- stair tolerance without stopping
- daily weight during periods of instability
- home blood pressure and heart rate logs, especially after medication changes
In CAV, early recognition of functional decline can be as valuable as a single test result, especially when it triggers timely imaging, immunosuppression review, and risk-factor tightening.
How CAV is diagnosed and monitored
CAV diagnosis is anchored in surveillance because symptoms may be late or nonspecific. Clinicians aim to detect disease early, classify severity, and identify whether epicardial disease, microvascular dysfunction, or both are limiting perfusion. Monitoring also helps separate CAV from other causes of graft dysfunction such as acute rejection, medication toxicity, or uncontrolled hypertension.
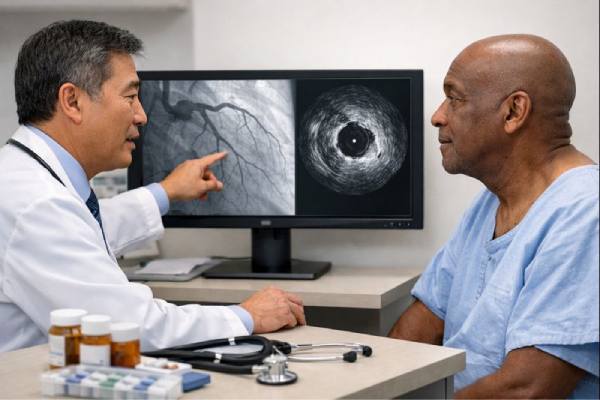
Coronary angiography as the traditional backbone
Invasive coronary angiography has long been the central test. It visualizes the larger epicardial vessels and can identify focal stenoses. Its limitation is that early CAV may be diffuse and concentric, which can appear deceptively “normal” if the entire vessel has narrowed uniformly.
To improve sensitivity, angiography is often combined with intracoronary imaging:
- Intravascular ultrasound (IVUS): Detects intimal thickening and plaque burden earlier than angiography alone and can track progression over time.
- Optical coherence tomography (OCT): Offers high-resolution plaque characterization in selected settings, though its role varies by center.
Many programs begin structured CAV screening around the first year post-transplant, then repeat testing at regular intervals based on individual risk, prior findings, and comorbidities. High-risk patients may be monitored more frequently, while lower-risk patients may shift to longer intervals or incorporate noninvasive approaches.
Noninvasive testing is expanding
Noninvasive tests are increasingly used to reduce repeated invasive procedures and to assess microvascular disease:
- Cardiac PET perfusion with myocardial blood flow measurement: Can quantify flow reserve and detect microvascular impairment that angiography might miss.
- Stress imaging (nuclear, echocardiography, or CMR in selected cases): Helps identify inducible ischemia and functional consequences.
- Coronary CT angiography: May be used in some patients for surveillance or problem-solving, though image quality and calcification, heart rate, and kidney function can affect suitability.
Biomarkers and functional clues
No blood test diagnoses CAV by itself, but clinicians sometimes use biomarkers to raise suspicion or track risk:
- troponin trends (especially high-sensitivity assays)
- natriuretic peptides in the context of graft dysfunction
- markers of inflammation or antibody activity when immune mechanisms are suspected
Staging and clinical decision-making
Monitoring is not only about “finding stenosis.” Clinicians interpret results through multiple lenses:
- Anatomic burden (angiography plus intracoronary imaging when available)
- Physiologic impact (flow reserve, perfusion defects, ventricular function)
- Clinical trajectory (exercise tolerance, arrhythmias, hospitalizations)
- Modifiers (rejection history, antibodies, infections, kidney function)
If test results and symptoms do not align, teams may reassess volume status, rhythm, anemia, and medication effects, and may repeat physiologic testing. The goal is a coherent explanation for how the graft is performing and what intervention—medical, procedural, or immunologic—is most likely to preserve function.
Treatment and long-term management
CAV treatment has two parallel aims: slow progression and manage established disease. Because CAV is diffuse and often involves microvasculature, the most effective strategy is usually prevention and early intervention rather than late mechanical rescue.
Foundational prevention strategies
Most transplant programs build CAV prevention into routine care from early after transplantation:
- Lipid lowering (often statin-based): Used broadly because it reduces lipid-driven endothelial injury and may also have anti-inflammatory benefits.
- Blood pressure control: Targets vary by patient, but stable control helps reduce endothelial stress and left ventricular workload.
- Glucose management: Tightening diabetes control reduces inflammatory signaling and vascular injury.
- Smoking avoidance and lifestyle support: Tobacco exposure is a strong vascular toxin; exercise and nutrition support improve metabolic resilience.
- Infection prevention: Vaccination, early infection treatment, and focused management of viral reactivation are often treated as part of vascular protection.
Immunosuppression optimization
Because immune injury is central to CAV biology, immunosuppression strategy is often reassessed when CAV appears or progresses:
- Ensuring consistent adherence and therapeutic drug levels
- Reviewing rejection history and antibody activity
- Considering regimen adjustments, including antiproliferative strategies in appropriate patients and settings
Some centers consider switching selected patients to regimens that may slow intimal hyperplasia, especially when progression is documented and risk-benefit balance is favorable. These decisions are individualized because medication changes can affect kidney function, infection risk, wound healing, lipid levels, and proteinuria.
Treating established CAV
When disease is clinically significant, management may include:
- Anti-ischemic and heart failure therapy: Diuretics, guideline-aligned heart failure medications as tolerated, and rhythm management when arrhythmias develop.
- Percutaneous coronary intervention (PCI): Useful for focal lesions, but outcomes can be limited by diffuse disease and restenosis risk. It is most effective when there is a clearly targetable stenosis causing ischemia.
- Surgical revascularization: Often challenging because of diffuse narrowing and small distal targets, and is used selectively.
- Advanced therapies: In severe, progressive disease with graft dysfunction where other options are exhausted, re-transplantation may be considered in carefully selected candidates.
What patients can do day to day
Long-term success depends on consistency:
- Take immunosuppressive medications exactly as prescribed and report missed doses promptly.
- Keep a personal record of key metrics: LDL, A1c (if diabetic), blood pressure averages, creatinine, and weight trends.
- Ask for a clear surveillance plan and understand what findings would change it.
- Report new exertional limits, palpitations, syncope, or unexplained swelling early rather than waiting for the next scheduled visit.
A practical takeaway: CAV is rarely controlled by one “big” intervention. It is controlled by persistent alignment—immune stability, infection control, and cardiovascular risk reduction—plus timely escalation when surveillance shows progression.
References
- The International Society for Heart and Lung Transplantation (ISHLT) guidelines for the care of heart transplant recipients – PubMed 2023 (Guideline)
- Cardiac allograft vasculopathy: current review and future research directions – PMC 2021 (Review)
- Cardiac Allograft Vasculopathy: Challenges and Advances in Invasive and Non-Invasive Diagnostic Modalities – PMC 2024 (Review)
- Cardiac Allograft Vasculopathy: A Focus on Advances in Diagnosis and Management – PMC 2025 (Review)
Disclaimer
This article is for educational purposes only and does not provide medical advice, diagnosis, or treatment. Cardiac allograft vasculopathy can be serious and may progress without typical chest pain, especially after heart transplantation. Seek urgent medical care for fainting, sudden or severe shortness of breath, chest pressure at rest, new rapid or irregular heartbeat with dizziness, or rapidly worsening swelling or weight gain. For personalized decisions about surveillance frequency, immunosuppression, lipid-lowering therapy, and procedural options, consult your transplant team or a qualified clinician who can review your symptoms, testing, and medication profile.
If you found this article helpful, please share it on Facebook, X (formerly Twitter), or any platform you prefer, and follow us on social media. Your support through sharing helps our team continue producing high-quality health content.






