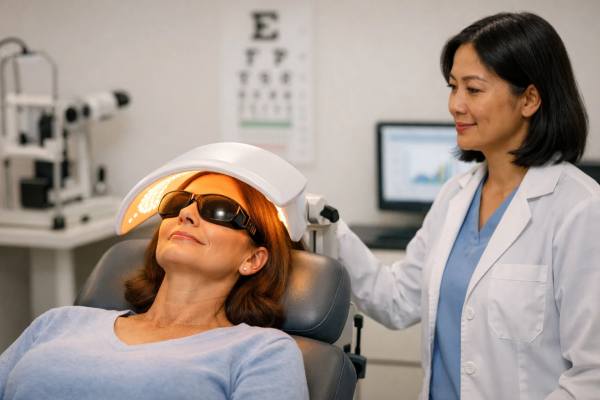
Dry eye is not just “not enough tears.” For many people, it is an unstable tear film driven by inflamed eyelids, thickened meibomian gland oil, and a surface that stays irritated despite good habits. Low-level light therapy (LLLT), also called photobiomodulation, has become a popular in-office option because it aims to calm inflammation and improve oil-gland function without cutting or injecting anything. Sessions are typically brief, noninvasive, and often paired with other eyelid treatments such as meibomian gland expression or intense pulsed light. What makes LLLT interesting is that it targets the biology underneath symptoms—cell energy pathways, blood flow signals, and inflammatory mediators—rather than simply replacing tears. Still, results vary by patient, device, and protocol, and the best outcomes usually come from matching the right person to the right plan. This article explains what LLLT is, how it may work, what clinical studies report, and how to think about safety, costs, and realistic expectations.
Quick Overview
- LLLT can improve dry eye symptoms and tear-film stability, especially in evaporative dry eye tied to meibomian gland dysfunction.
- Best results are usually seen after a short series of sessions rather than a single visit, with benefits that may fade without maintenance.
- Evidence is strongest for in-office systems; at-home red-light devices often lack validated dosing and eye-safety safeguards.
- People with active eye infection, certain photosensitivity conditions, or uncontrolled eyelid inflammation may need treatment delays or alternatives.
- A practical starting point is a 3–6 week treatment series, followed by a reassessment using symptom scores and tear break-up time.
Table of Contents
- What low-level light therapy is and is not
- How photobiomodulation may help dry eye
- What clinical studies report so far
- Who tends to benefit most
- Treatment protocols and what to expect
- Side effects and safety considerations
- How LLT fits into a complete dry eye plan
What low-level light therapy is and is not
Low-level light therapy for dry eye is a form of medical phototherapy that uses low-energy light—most commonly in the red to near-infrared range—to influence cellular activity. You may also hear it called photobiomodulation. In practical terms, it is usually delivered with a mask or panel placed near the eyes while your lids are closed, sometimes alongside warm eyelid treatment or other light-based therapy.
It helps to separate LLLT from other “light treatments,” because the names can blur together.
- LLLT or photobiomodulation: low-intensity light intended to trigger biological signaling rather than to heat tissue aggressively or destroy structures.
- Intense pulsed light: high-energy pulses applied to the skin around the eyelids, traditionally used in dermatology and now commonly used for meibomian gland dysfunction and ocular rosacea patterns.
- Low-level light plus intense pulsed light: combination protocols are common in dry eye clinics; the two are different technologies with different targets.
Dry eye itself is also not a single disease. LLLT is usually discussed in the context of evaporative dry eye, where the main problem is poor-quality oil from the meibomian glands. Those glands line the eyelids and supply the outer lipid layer of the tear film. When oil is too thick or glands are inflamed, tears evaporate faster and the surface becomes irritated even if tear volume is normal.
LLLT is not a stand-alone “cure” for every type of dry eye. If your primary issue is aqueous deficiency (low tear production), autoimmune disease, medication-related dryness, or exposure problems from incomplete blinking, light therapy may be supportive but rarely sufficient by itself. It is also not an instant fix: many protocols are designed as a series, and the best measure of success is not only how you feel in the chair that day, but what happens to symptoms, tear stability, and gland function over the following weeks.
A useful mindset is this: LLLT is an in-office tool that aims to make the eyelids and tear film more stable, so that your daily treatments—lubrication, lid hygiene, anti-inflammatory drops, and environmental control—work better and feel easier to sustain.
How photobiomodulation may help dry eye
Dry eye symptoms often come from a feedback loop: unstable tears irritate the surface, irritation drives inflammation, inflammation worsens gland function, and glands produce poorer oil that makes tears even less stable. Photobiomodulation is appealing because it targets multiple parts of that loop at once.
At the cellular level, red and near-infrared light is thought to interact with mitochondrial pathways—the “energy factories” of cells. In many tissues, low-level light exposure has been associated with changes in cellular energy availability, oxidative stress signaling, and inflammatory mediator balance. In the eyelids, those shifts may support healthier gland cell activity and calmer inflammatory behavior.
For evaporative dry eye, the most relevant targets include:
- Meibomian gland function: improving the quality and expressibility of meibum (the oil that should be clear and flow easily).
- Eyelid inflammation: reducing inflammatory cytokine signaling that contributes to redness, irritation, and gland blockage.
- Microcirculation: supporting local blood flow signals that may improve tissue recovery and reduce chronic congestion in lid margins.
- Nerve sensitivity: some patients notice burning and light sensitivity that behave like an “irritated nerve” state; calming surface inflammation can reduce that sensitization over time.
It is also important to address the common misconception that LLLT is simply “heating the glands.” Some systems may raise eyelid temperature modestly, which can help soften thick oil, but photobiomodulation is generally described as a signaling-based therapy rather than a purely thermal one. That distinction matters because it influences expectations. Heat can produce short-term relief, but signaling changes may take repeated exposures to translate into more stable symptoms.
Many clinics combine LLLT with a hands-on eyelid step such as meibomian gland expression. That pairing makes intuitive sense: light may help reduce inflammatory load and improve oil properties, while expression physically clears gland obstruction. When patients only do one piece of that puzzle, improvements can be smaller or shorter-lived.
A practical way to think about mechanism is to focus on what you can measure:
- Symptom scores: how often and how severely symptoms interfere with reading, screens, driving, and comfort.
- Tear break-up time: how quickly the tear film becomes patchy after a blink.
- Lid margin findings: redness, thickened secretions, blocked orifices, and gland dropout patterns.
If LLLT is working, you often see a combination of better symptoms and at least one objective sign moving in the right direction. If symptoms improve but objective signs do not, it may still be meaningful, but your clinician should consider whether other drivers—such as allergy, neuropathic pain, or incomplete blinking—are dominating the picture.
What clinical studies report so far
Research on LLLT for dry eye has expanded quickly, but it is not perfectly uniform. Different studies use different wavelengths, treatment times, and companion therapies. That makes “one-size-fits-all” conclusions unreliable. Still, patterns have emerged.
In stand-alone LLLT studies, investigators commonly report improvements in symptoms and tear stability measures after a short series of treatments. A typical protocol in published work ranges from three visits over a few weeks to six sessions over about three weeks, with follow-up commonly at one to three months. Improvements often include:
- Lower symptom scores, frequently measured by standardized questionnaires.
- Longer tear break-up time, meaning the tear film remains smooth for longer after blinking.
- Better lipid layer behavior or meibum quality measures in patients with meibomian gland dysfunction.
- Minimal change in structural gland loss over short timeframes, which is expected because gland architecture does not rebuild quickly.
Combination therapy studies—particularly LLLT paired with intense pulsed light—tend to show stronger or more consistent improvements in some outcomes, especially for patients with moderate to severe meibomian gland dysfunction or rosacea-associated eyelid disease. When combination therapy works well, patients often report fewer “bad eye days,” less burning, and improved visual comfort during screens. Objective measures such as tear break-up time can improve by a few seconds on average in many reports, and symptom score reductions are often clinically noticeable.
Two details are worth emphasizing because they help set expectations.
Benefits are often front-loaded
Many patients notice some symptom relief early, sometimes after the first or second session. But the more reliable goal is a smoother baseline after the full series. If you judge success after a single visit, you may miss the cumulative effect that the protocol is designed to create.
Durability varies
Some patients maintain improvement for months, while others gradually drift back as glands clog again or inflammation flares. This is not necessarily a “failure.” It can mean that the underlying disease is still active and needs either maintenance sessions or stronger daily control (lid hygiene, anti-inflammatory drops, allergy management, blink training, or treatment of ocular rosacea drivers).
The best research also reinforces a practical clinical point: dry eye outcomes depend heavily on selecting the right population. When studies enroll mostly evaporative dry eye patients with meibomian gland dysfunction, results look more favorable than when they include mixed dry eye types without clear gland-related criteria.
If you are considering LLLT, ask your clinician which outcomes they track in their practice. A good clinic will measure more than comfort alone, such as tear break-up time, lid margin quality, and gland expressibility. Those objective markers make it easier to decide whether LLLT should be repeated, paired with another therapy, or replaced with a different approach.
Who tends to benefit most
LLLT is most often offered to patients whose dry eye is driven by meibomian gland dysfunction. The simplest way to recognize that pattern is to look for evaporative features: symptoms worse late in the day, gritty burning that flares with air flow, and vision that “smears” after long screen sessions but improves after blinking. Clinicians may also see thickened oil, capped gland openings, telangiectasia along the lid margin, and rapid tear break-up time.
People who often do well include:
- Patients with moderate meibomian gland dysfunction who have tried consistent warm compresses and lid hygiene but still have unstable tears.
- Patients with ocular rosacea patterns, where inflammation and vascular changes contribute to gland dysfunction.
- Contact lens wearers who have dryness driven by gland instability, especially if lenses become uncomfortable late in the day.
- Patients who cannot tolerate some topical medications or who want to reduce reliance on frequent drops.
LLLT may be less helpful, or require a different primary plan, when the main driver is not the eyelid oil glands. Examples include:
- Predominantly aqueous-deficient dry eye, where tear production is low and gland issues are secondary.
- Significant allergic conjunctivitis, where itch and seasonal flares dominate symptoms.
- Exposure-related dryness from incomplete blinking, eyelid laxity, or nocturnal lagophthalmos.
- Neuropathic ocular pain, where pain is disproportionate to surface findings and needs a broader nerve-focused plan.
It is also reasonable to be cautious if your dry eye is in a highly inflamed phase. If the ocular surface is very irritated, your clinician may first calm things with anti-inflammatory drops, allergy control, lid margin treatment, or punctal management before adding light-based therapy. Starting LLLT too early in an unstable flare can lead to disappointment because the underlying inflammatory load is too high to see the true effect.
Key screening questions to ask
- Do I have meibomian gland dysfunction, and how severe is it?
- Is my dry eye primarily evaporative, aqueous-deficient, or mixed?
- What objective measures will we track before and after treatment?
- Will you do meibomian gland expression or another lid procedure as part of the plan?
- If I improve, what is the expected maintenance strategy?
A thoughtful match between the treatment and your dry eye subtype is often the difference between “it helped a little” and “it noticeably changed my baseline.” When LLLT is used in the right context, it can become a durable part of a long-term plan rather than a one-off procedure.
Treatment protocols and what to expect
LLLT protocols vary by device and clinic, but most follow a series-based model. The most common schedules fall into one of these patterns:
- Short intensive series: 2 sessions per week for 3 weeks, totaling 6 treatments
- Weekly series: 3 sessions over 3 weeks
- Spaced series: 4 sessions spaced about every 2 to 3 weeks
- Combination series: intense pulsed light sessions with LLLT performed the same day or shortly after
During a session, you typically sit or recline with your eyes closed. The device may be a mask or panel delivering red or near-infrared light near the eyelids. Most patients report warmth or a gentle heating sensation rather than pain. If you have strong light sensitivity, tell the clinician in advance so they can confirm protective measures and adjust the environment.
Clinics often pair LLLT with other steps that matter as much as the light itself:
- Meibomian gland expression: gentle pressure on eyelids to clear thickened oil
- Lid margin debridement: cleaning debris and biofilm that blocks gland openings
- Warm compress or heat-based devices: softening oil before expression
- Skin-directed therapy: addressing rosacea-related inflammation in the periocular area
How quickly does it work?
Many patients notice reduced burning or improved comfort within 1 to 2 sessions, but the steadier change often appears after completing the full series. Vision quality during screens may also improve as tear stability increases, but that can lag behind comfort improvements by a week or two.
How long do results last?
Durability is variable. A practical range many clinicians discuss is 3 to 9 months of benefit, with some patients maintaining improvement longer when they keep up home care and treat contributing factors such as allergy or ocular rosacea. Others need maintenance sessions. The most useful approach is to treat LLLT as a “reset” that creates a better baseline, then protect that baseline with consistent daily measures.
What should be measured afterward?
A strong follow-up visit includes both symptoms and objective checks:
- Symptom score trend compared with baseline
- Tear break-up time
- Meibum quality and expressibility
- Lid margin inflammation and debris
- Corneal staining if the surface was previously irritated
If you do not improve after a full series, the next step is not simply “do more sessions.” It is to reassess the diagnosis. Persistent symptoms after well-delivered LLLT often mean another driver is present—aqueous deficiency, allergy, exposure, or neuropathic pain—or that companion treatments were missing.
Side effects and safety considerations
LLLT for dry eye is generally described as well tolerated when delivered by trained professionals using devices designed for periocular use. Most reported side effects are mild and temporary, but safety still deserves careful attention because the eyes are light-sensitive organs.
Common short-term effects can include:
- Mild warmth, eyelid redness, or facial flushing that resolves within hours
- Temporary dryness or irritation the same day, especially if glands are expressed afterward
- Headache or light sensitivity in people prone to migraine
- Temporary blur from tear-film changes immediately after treatment
More significant problems are uncommon, but clinicians should screen for contraindications and use protective measures.
Situations that may require postponing treatment
- Active eye infection, such as conjunctivitis, styes with significant inflammation, or active blepharitis flare
- Recent ocular surgery or procedures where your surgeon advised avoiding periocular treatments
- Uncontrolled ocular surface inflammation with significant corneal staining that needs stabilization first
- Active skin lesions in the treatment area that require medical evaluation
Photosensitivity and medication cautions
Some medical conditions and medications increase photosensitivity. Even though LLLT uses low energy, clinics may avoid treatment or modify protocols if you have a known photosensitivity disorder, a history of light-triggered seizures, or you are taking medications that make your skin unusually light-sensitive. This is not a reason to panic; it is a reason to disclose your full medication list and medical history so your clinician can make a safe call.
At-home devices need extra skepticism
Consumer “red light masks” are widely sold, but many are not designed for safe use near the eyes, and dosing can be unclear. Eye safety is not only about wavelength; it is also about irradiance, exposure time, distance, and how well the device prevents direct light entry. If you are considering an at-home product, treat it as a medical decision: ask whether it has credible periocular safety testing and whether the manufacturer provides clear dosing and protective guidance. If those elements are missing, it is safer to avoid improvising.
The most important safety rule is simple: new severe pain, significant vision change, or persistent redness after any eye procedure should be evaluated promptly. While these outcomes are not expected with proper LLLT, quick assessment is always the right move when vision is involved.
How LLT fits into a complete dry eye plan
LLLT is best viewed as one part of a layered strategy. Dry eye is chronic for many people, and the most reliable improvement usually comes from combining in-office “boosters” with daily maintenance that fits real life.
A practical framework is to treat three pillars: tear film, eyelids, and inflammation.
Tear film support
- Preservative-free lubricating drops used consistently rather than only when symptoms spike
- Gel drops or ointment at night if morning dryness or nocturnal exposure is present
- Humidity and airflow control, especially for screens, commuting, and sleep
Eyelid and meibomian gland support
- Daily warm compresses that are truly warm for 8 to 10 minutes
- Gentle lid cleansing if debris or biofilm is present
- Blink training for heavy screen users, focusing on complete blinks rather than frequent partial blinks
- In-office expression or thermal pulsation in selected cases when glands remain obstructed
Inflammation control
- Allergy management when itch and seasonal flares are part of the pattern
- Anti-inflammatory prescription drops when inflammation is persistent or corneal staining is present
- Targeted management of ocular rosacea drivers when lid margin blood vessels and facial flushing are involved
Where does LLLT fit? It often works as a catalyst that improves gland function and lowers inflammatory tone enough that your daily routine becomes easier and more effective. It can also be a useful option when intense pulsed light is contraindicated or poorly tolerated, although many clinics still favor combination protocols for more stubborn meibomian gland dysfunction.
Questions that protect you from overpaying
- What is the plan if I do not respond after the first series?
- Will you re-measure tear break-up time and meibum quality, or are we judging only by symptoms?
- How do you decide on maintenance timing—symptoms, exam findings, or both?
- What home plan should I follow during the series to protect results?
Finally, be careful with expectations. LLLT is not usually a replacement for every other therapy. It is most often a way to reduce the “noise” of constant irritation so that other treatments can actually hold. When it is used thoughtfully—based on dry eye subtype, objective findings, and a clear follow-up plan—it can meaningfully improve comfort, vision quality, and daily function.
References
- Low-level light therapy alone versus combination therapy with intense pulsed light in the treatment of dry eye disease with meibomian gland dysfunction: A randomised paired-eye and mechanism of action trial 2025 (RCT)
- Low-level light therapy and intense pulse light therapy in meibomian gland dysfunction. A systematic review and meta-analysis 2024 (Systematic Review)
- A randomized controlled study evaluating outcomes of intense pulsed light and low-level light therapy for treating meibomian gland dysfunction and evaporative dry eye 2023 (RCT)
- Effect of low-level light therapy in patients with dry eye: a prospective, randomized, observer-masked trial 2022 (RCT)
- Effect of low-level light therapy in individuals with dry eye disease 2024 (Clinical Study)
Disclaimer
This article is for educational purposes only and does not provide medical advice, diagnosis, or treatment. Dry eye has multiple causes, and light-based therapies may be inappropriate for certain eye conditions, skin conditions, or medication-related photosensitivity risks. If you have severe eye pain, sudden vision changes, significant redness, or new light sensitivity after any procedure, seek prompt evaluation by a licensed eye care professional. For personalized guidance, schedule an exam with an optometrist or ophthalmologist who can assess your tear film, eyelids, and ocular surface and recommend a plan tailored to your health history.
If you found this article useful, please consider sharing it on Facebook, X (formerly Twitter), or any platform you prefer.