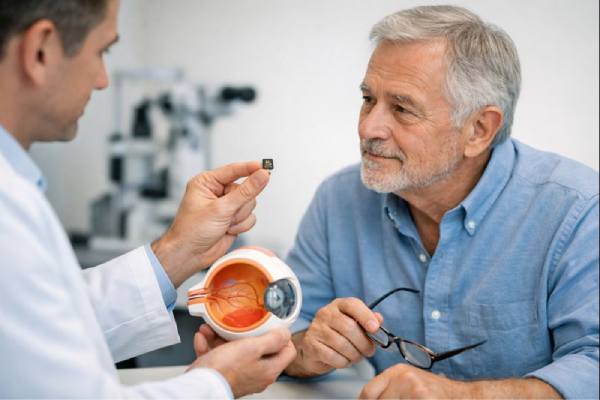
For people with advanced dry age-related macular degeneration (AMD), the hardest loss is often central vision—the ability to read, recognize faces, and see fine detail straight ahead. Subretinal retinal prosthesis implants are a new approach designed for that exact problem. Instead of trying to revive dead photoreceptors, a tiny “chip” placed under the atrophic macula converts patterned light into electrical signals that stimulate surviving retinal neurons. When paired with special glasses that project near-infrared images onto the implant, some patients can regain a form of central “pattern” vision while keeping their natural peripheral vision.
This is not a cure, and it is not the same as restoring youthful eyesight. The best results depend on careful selection, realistic expectations, and training to learn how prosthetic vision behaves. Still, the concept is notable: it aims to restore usable central vision in a condition where most therapies focus on slowing further loss. This article explains how subretinal chips work, who may benefit, what outcomes are realistic, and the key risks to understand.
Key Insights
- Subretinal chips can provide a new, low-resolution form of central vision in selected people with geographic atrophy who have lost foveal function but retain peripheral vision.
- The system relies on external glasses and structured rehabilitation; gains often improve with practice, magnification strategies, and task-specific training.
- It remains a specialized surgical option with meaningful risks, including inflammation, retinal complications, and the possibility of limited benefit.
- A practical next step is to seek evaluation at a retinal center experienced in advanced AMD and low-vision rehabilitation to discuss candidacy and access pathways.
Table of Contents
- Why subretinal chips fit dry AMD
- How the chip and glasses create vision
- Who may benefit and who should not
- What central vision restoration really means
- Surgery, recovery, and training timeline
- Risks, complications, and long-term questions
Why subretinal chips fit dry AMD
Dry AMD becomes disabling when it progresses to geographic atrophy (GA), where the photoreceptors and supporting retinal pigment epithelium in the macula progressively die. The central scotoma that follows is not just “blur.” It is missing information—letters vanish, faces become unrecognizable, and straight-ahead detail collapses even when side vision remains.
That pattern—severe central loss with preserved peripheral navigation—is exactly what makes subretinal prostheses conceptually attractive. Many people with GA still have functioning inner retinal neurons (such as bipolar and ganglion cells) outside or around the atrophic zone, and the optic nerve can still carry signals to the brain. A subretinal implant aims to substitute for lost photoreceptors in the damaged central region by directly driving the remaining retinal circuitry.
Why “subretinal” placement matters
Subretinal chips sit where photoreceptors used to be, beneath the retina in the region of atrophy. This positioning has practical implications:
- The stimulation can be more “retina-native,” leveraging local retinal processing rather than bypassing it entirely.
- The implant can be confined to the central atrophic area, so patients can potentially use prosthetic central vision together with natural peripheral vision.
- Because GA eyes often retain peripheral function, the goal is not to replace the whole visual field—only to reintroduce central pattern information for tasks like letter recognition.
Why this differs from slowing GA
Many dry AMD interventions aim to slow progression. That can be valuable, but it does not restore what has already been lost. Subretinal prosthesis systems are aimed at a different need: individuals who already have profound central vision loss and want functional improvement, even if limited in resolution.
Where expectations must be disciplined
Even in ideal candidates, the “restored” vision is typically monochromatic, low-resolution, and dependent on the external imaging system. It is closer to regaining a workable central signal than returning to normal sight. A helpful mental model is: the system may give you a new way to read large print, identify high-contrast objects, and orient within a scene—especially with magnification and training—while your peripheral vision continues to handle navigation.
How the chip and glasses create vision
Subretinal chip systems for dry AMD work as a paired platform: an implanted microchip under the macula plus wearable glasses that deliver patterned light to that chip. The chip is “wireless” in the everyday sense—there is no cable exiting the eye—yet it still needs energy and visual information. Both are delivered through light.
The basic signal path
- A camera on the glasses captures the scene in front of you.
- A small processor enhances the image (for example, contrast, edge emphasis, or magnification).
- The glasses project a patterned near-infrared image onto the retina, aligned to the implant area.
- Pixels in the chip convert that light into tiny electrical currents.
- Those currents stimulate nearby retinal neurons, and the signal is relayed through the optic nerve to the brain.
Near-infrared projection is used so the projected image can drive the implant without competing with visible-light input that supports peripheral natural vision. In practical terms, it helps separate the “prosthetic central channel” from what your remaining retina still sees.
Pixels, resolution, and why magnification is central
A chip’s pixel size imposes a hard ceiling on detail. If pixels are spaced widely, the visual system cannot represent fine lines or small letters without magnification. That is why these systems typically rely on zoom: the processor enlarges a letter or object so it spans multiple pixels, making it recognizable.
This is also why “restoring central vision” often looks like a partnership between technology and technique. The device provides a stable, repeatable stimulus; the patient learns how to use magnification, scanning, and positioning to extract information efficiently.
Why rehabilitation is part of the mechanism
With natural vision, your brain seamlessly fuses detail and context. With a subretinal prosthesis, the central image has a different texture and a different relationship to head movement, contrast, and distance. Many people need structured practice to become fluent—similar to learning a new interface. Training often focuses on:
- Fixation strategy: keeping the target aligned with the projected area
- Scanning: controlled head and eye movements to explore shapes
- Contrast management: choosing lighting and backgrounds that “read” well
- Task progression: from simple high-contrast shapes to letters and then to words
What the system is not doing
It is not regrowing photoreceptors. It is not reversing GA. And it is not giving back high-definition color vision. The system is providing an alternative way to deliver patterned information to surviving retinal neurons so the brain can reconstruct a useful central percept—often best for high-contrast, structured tasks.
Who may benefit and who should not
Candidacy is the make-or-break step. Subretinal chips are intended for a narrow clinical situation: advanced central loss from GA with enough remaining retinal and optic nerve function to transmit a meaningful signal. The evaluation is typically more detailed than a standard “AMD check,” because the goal is not diagnosis—it is predicting whether a prosthetic signal can be used.
Common features of good candidates
While exact thresholds vary by program and protocol, many suitable candidates share these traits:
- Advanced dry AMD with a well-defined central atrophic zone involving the fovea
- Profound central vision loss that prevents functional reading with the affected eye
- Preserved peripheral vision that supports mobility and spatial orientation
- Retinal anatomy that can safely accept a subretinal implant (assessed with imaging)
- Motivation for training and willingness to work through a learning curve
In real life, “motivation” is not a vague personality trait. It shows up as patience with practice sessions, willingness to use the glasses consistently, and interest in task-based goals (reading labels, identifying numbers, large-print text).
Factors that can limit benefit
Several issues can reduce the likelihood of meaningful functional gain:
- Extensive retinal scarring or distortion that prevents stable chip positioning
- Significant optic nerve disease or advanced glaucoma that reduces signal transmission
- Macular problems beyond GA that degrade the inner retina’s ability to respond
- Severe fixation instability that prevents consistent alignment of the projected image
- Cognitive or neurologic barriers that make training and integration difficult
When a prosthesis may be inappropriate
Subretinal prostheses are not a first-line option for earlier dry AMD where central vision is still usable. They also may not be appropriate when:
- The better eye still provides strong central function and the surgical risk feels disproportionate
- There is active ocular infection or uncontrolled inflammation
- The eye has structural risks that make retinal surgery unusually hazardous
- Expectations are anchored to “normal vision” rather than functional improvement
Why “one eye at a time” is common
Because GA is often bilateral, people may wonder about treatment in both eyes. Many programs prioritize one eye—often the worse-seeing eye—so the person retains a stable reference eye for daily function. This also allows the brain to integrate prosthetic central input while still relying on the other eye’s natural vision, which can ease adaptation.
What central vision restoration really means
The phrase “restore central vision” can be misleading if it is heard as “restore normal sight.” A better phrasing is: subretinal chips can restore formed central percepts—structured visual information that can be trained and used for specific tasks. The difference matters because it sets expectations for daily life.
Typical strengths of prosthetic central vision
In successful users, the most common functional wins tend to be:
- Recognizing large letters and numbers, especially with magnification
- Identifying high-contrast shapes and boundaries (door frames, bold signs)
- Locating objects on a tabletop with controlled scanning
- Combining peripheral natural vision (navigation) with central prosthetic input (detail)
Many patients describe prosthetic vision as “workable” rather than “effortless.” It can reduce dependence on guesswork and make structured tasks less frustrating, but it usually remains slower than natural vision.
Why reading is possible but not automatic
Reading with a prosthesis is often a staged skill:
- Single letters at high contrast and large size
- Short sequences (two to four letters)
- Word recognition with repeated practice and stable viewing distance
- Functional reading tasks (labels, headings, short messages)
Magnification is central. Instead of expecting fine print, the user learns to scale text and control distance so the letter fills enough pixels to be distinguishable.
What remains difficult
Even with good outcomes, certain tasks tend to remain challenging:
- Face recognition at conversational distance, especially in low contrast
- Fast reading of continuous text
- Low-light scenes, glare, and complex visual clutter
- Color-dependent tasks, because prosthetic vision is typically monochromatic
This is where practical planning helps. A realistic goal is not “I will read novels again” but “I may reliably read large print and labels, and I can build strategies for more.”
How to judge success thoughtfully
Success should be measured across three levels:
- Clinical performance: letter recognition or visual acuity metrics under standardized conditions
- Functional performance: what you can do at home (mail, medicine labels, appliance settings)
- Burden of use: comfort with the glasses, fatigue, and how often you choose to use prosthetic vision
A meaningful outcome is one where functional gains outweigh the effort and the device becomes a tool you actually reach for, not a system that sits unused because the workflow feels too demanding.
Surgery, recovery, and training timeline
Subretinal implantation is delicate retinal surgery. Even though the device is tiny, placing it under the retina requires controlled steps, and recovery is a process—not a single event. Understanding the timeline helps people prepare emotionally and practically.
Before surgery: stabilization and mapping
Preoperative work typically includes detailed retinal imaging to map the atrophic zone, confirm peripheral retinal health, and evaluate whether the retina can be safely lifted and reattached. The team may also assess fixation behavior and discuss whether the chosen eye and scotoma pattern are likely to support training.
You should expect a frank conversation about goals. Many programs encourage patients to define two or three practical targets—such as reading large-print labels, recognizing numbers, or improving the ability to locate objects—because those targets guide training later.
The procedure in broad strokes
While exact technique varies, implantation often involves:
- Vitrectomy to access the retina safely
- Creating a controlled retinal detachment in the target area
- Placing the chip under the retina in the atrophic macula
- Reattaching the retina so it settles over the implant
- Managing the eye’s internal environment to support healing
The procedure aims to keep peripheral vision intact while introducing a new central input zone. That balance is a major reason candidate selection is strict.
Early recovery: healing before performance
In the first weeks, the priority is anatomical stability—retinal attachment, inflammation control, and monitoring for complications. During this phase, visual performance may be inconsistent. Some people notice initial percepts early; others need more healing time before the system can be activated and used reliably.
Activation and training: where gains are built
Once the eye is stable, rehabilitation begins. Training often includes:
- Learning alignment between the camera, projector, and implant zone
- Practicing high-contrast targets at a fixed distance
- Building scanning habits that reduce confusion and fatigue
- Introducing magnification workflows for letters and numbers
- Translating clinic skills into home tasks with coaching
Progress often comes in “plateaus and jumps.” It is common to feel stalled, then suddenly improve when a scanning strategy clicks or when magnification settings are optimized.
Long-term follow-up: adaptation and refinement
Over months, many users refine how they deploy prosthetic vision—when to use it, what lighting works best, and which tasks benefit most. Follow-up also monitors device positioning, retinal health, and any secondary issues that might affect function.
The practical takeaway is that surgery creates the possibility of restored central input, but rehabilitation turns it into real-life function.
Risks, complications, and long-term questions
Because subretinal chips involve intraocular surgery, the risk profile is closer to complex retinal procedures than to noninvasive vision aids. A balanced view acknowledges two truths at once: serious complications are not common in experienced hands, and when they occur, they can be vision-threatening.
Surgical and ocular risks
Potential risks include:
- Infection inside the eye, which requires urgent treatment
- Retinal detachment or re-detachment
- Bleeding inside the eye
- Persistent inflammation or scarring that affects retinal function
- Macular changes that reduce the quality of either prosthetic or residual natural vision
- Worsening of existing eye conditions that complicate recovery
Even when the implant is aimed at an already nonfunctional central area, peripheral vision protection remains a priority, so any complication that spreads beyond the implant zone matters.
Device-related and system risks
Because the implant is part of a system, challenges can arise at multiple points:
- Misalignment or stability issues that make percepts inconsistent
- Limited benefit due to biological factors, despite technically successful surgery
- Hardware workflow barriers, such as comfort with the glasses or fatigue with prolonged use
- Rehabilitation drop-off if training is not accessible or not sustained
A subtle risk is “functional non-use”: the device works in testing but does not integrate into daily life because the effort-to-benefit ratio feels unfavorable. That outcome is less dramatic than a surgical complication, but it matters deeply to patients.
Longevity and upgrade questions
Long-term questions include how the implant and surrounding retina behave over years, how durable functional gains are, and what happens if technology improves. Some research suggests that removal or replacement strategies may be possible in principle, but any additional surgery carries its own risk and must be justified by meaningful expected benefit.
Where this fits in the broader AMD landscape
Subretinal prostheses are best viewed as a targeted option for people with profound central loss who want potential restoration, not just slowing of decline. They do not replace low-vision services, and they do not eliminate the need for AMD monitoring. Instead, they add a new category of possibility: a central “signal” that can be trained, used, and combined with remaining peripheral vision.
If you are considering this path, the most protective step is a comprehensive evaluation at a center experienced in both advanced AMD and retinal surgery, with a rehabilitation plan that is as concrete as the surgical plan.
References
- Subretinal Photovoltaic Implant to Restore Vision in Geographic Atrophy Due to AMD – PubMed 2025 (RCT)
- Prosthetic Visual Acuity with the PRIMA Subretinal Microchip in Patients with Atrophic Age-Related Macular Degeneration at 4 Years Follow-up – PMC 2024 (Clinical Follow-up)
- Simultaneous perception of prosthetic and natural vision in AMD patients – PubMed 2022 (Clinical Trial)
- Enhancing Prosthetic Vision by Upgrade of a Subretinal Photovoltaic Implant in situ – PMC 2024 (Translational Study)
Disclaimer
This article is for general educational purposes and does not provide medical advice. Retinal prosthesis implantation for dry AMD is a complex, highly specialized intervention that may be available only in select centers and may be limited to specific eligibility criteria. Whether it is appropriate depends on detailed retinal imaging, overall eye health, medical history, and a careful discussion of benefits, limitations, and potential complications. Any intraocular surgery carries risks, including rare but serious events that can permanently worsen vision. Seek urgent eye care for sudden vision loss, flashes of light, a curtain-like shadow, severe eye pain, rapidly increasing redness, or new discharge.
If you found this article helpful, please consider sharing it on Facebook, X (formerly Twitter), or any platform you prefer.